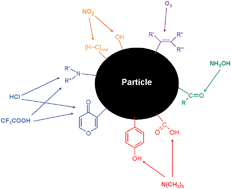
Six gases [N(CH3)3, NH2OH, CF3COOH, HCl, NO2 and O3] were selected to probe the surface of seven different types of combustion aerosol samples (amorphous carbon, flame soot) and three types of TiO2nanoparticles using heterogeneous, i.e. gas–surface reactions. The gas uptake to saturation of the probes was measured under molecular flow conditions in a Knudsen flow reactor and expressed as a density of surface functional groups on a particular aerosol, namely acidic (carboxylic) and basic (conjugated oxides such as pyrone, N-heterocycle and amine) sites, carbonyl (R1–C(O)–R2) and oxidizable (olefinic, –OH) groups. The limit of detection was generally well below 1% of a formal monolayer of adsorbed probe gas. With few exceptions most investigated aerosol samples interacted with all probe gases to various extents which points to the coexistence of different functional groups on the same aerosol surface such as acidic and basic groups. Generally, the carbonaceous particles displayed significant differences in surface group density: Printex 60 amorphous carbon had the lowest density of surface functional groups throughout, whereas Diesel soot recovered from a Diesel particulate filter had the largest. The presence of basic oxides on carbonaceous aerosol particles was inferred from the ratio of uptakes of CF3COOH and HCl owing to the larger stability of the acetate compared to the chloride counterion in the resulting pyrylium salt. Both soots generated from a rich and a lean hexane diffusion flame had a large density of oxidizable groups similar to amorphous carbon FS 101. TiO2 15 had the lowest density of functional groups studied for all probe gases among the three TiO2nanoparticles despite the smallest size of its primary particles. The technique used enabled the measurement of the uptake probability of the probe gases on the various supported aerosol samples. The initial uptake probability, γ0, of the probe gas onto the supported nanoparticles differed significantly among the various investigated aerosol samples but was roughly correlated with the density of surface groups, as expected.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...