Topological study of intramolecular hydrogen bonding in β-hydroxyethylperoxy radical and β-hydroxyethoxy radical along its dissociation pathway
Abstract
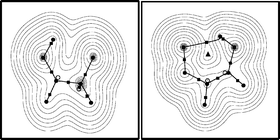
The presence of intramolecular hydrogen bond (IHB) in β-hydroxyethylperoxy and β-hydroxyethoxy radicals was investigated using the QTAIM topological study on UB3LYP/6-311++G(2d,2p) charge densities. Only one of the two conformers of β-hydroxyethylperoxy radical which were previously considered to present IHB displays a bond critical point (BCP) associated to an IHB. Furthermore, the atomic energies and electron populations indicate no evidence of IHB in the second conformer. Nevertheless, very small differences in molecular energies were obtained using several one-step and multi-step methods (G3, G3B3) between both conformers. No BCP is found between the hydroxyl hydrogen and the oxygen in the most stable conformer or in the transition state for the dissociation path of β-hydroxyethoxy radical. However, a BCP is formed in the last steps of this path, thereby yielding H-bonded products.

 Please wait while we load your content...
Please wait while we load your content...