Superior adsorptive removal of anionic dyes by MIL-101 analogues: the effect of free carboxylic acid groups in the pore channels†
Abstract
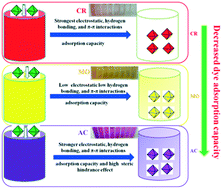
We herein report that the adsorption driving forces (i.e., electrostatic interactions, hydrogen bonding, and π–π interactions) of MIL-101-COOH microcrystals toward dyes can be tuned by varying the molar ratio of H2BDC : H2BDC–COOH in the reaction system. More specifically, the adsorption capacities of MIL-101-COOH-1 were determined to be 2835.7 (Congo red, CR), 473.9 (methyl orange, MO), and 240.8 (acid chrome blue K, AC) mg g−1, exceeding those reported for pristine MIL-101 by 107.5, 16.7, and −25.8%, respectively. The above-mentioned adsorption behavior was rationalized based on the dye molecular structure, zeta potential, BET surface area and dye adsorption investigations, which revealed that free –COOH groups increase the electrostatic and hydrogen bonding interactions between the adsorbent and linear dyes, thereby improving the adsorption capacity for linear anionic dyes and overriding the effect of pore volume, whereas an increased steric hindrance effect between the adsorbent and a nonlinear dye decreases the adsorption capacity for the nonlinear anionic dye and overrides the effects of the electrostatic and hydrogen bonding interactions. The adsorption capacity of MIL-101-COOH-1 toward the anionic organic dyes (CR, MO, and AC) was found to be significantly higher than those of the other MIL-101-COOH adsorbents examined herein. The adsorption capacity of MIL-101-COOH is determined by the synergetic interplay between the electrostatic, hydrogen bonding, and π–π interactions, steric hindrance effect, and pore volume of the adsorbent. The results shed light on the adsorption mechanism of metal–organic frameworks toward organic dyes.


 Please wait while we load your content...
Please wait while we load your content...