Does stoichiometry matter? Cocrystals of aliphatic dicarboxylic acids with isonicotinamide: odd–even alternation in melting points†
Abstract
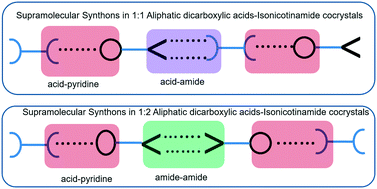
This study outlines the synthesis of four cocrystals of aliphatic dicarboxylic acids {pimelic acid to sebacic acid (HOOC–(CH2)n–COOH, n = 5, 6, 7 and 8)} and isonicotinamide in a ratio of 1 : 2. Further, the importance of stoichiometry of chemical components and complementary hydrogen bonds is emphasized by CSD analysis of 14 different cocrystals. Subsequently, the odd–even alternation in melting points of dicarboxylic acid and isonicotinamide cocrystals is studied. Cocrystals containing even dicarboxylic acids (diacids) show higher melting points than odd diacids.


 Please wait while we load your content...
Please wait while we load your content...