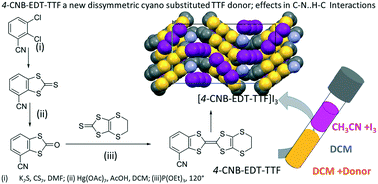
A 4-cyanobenzene-ethylenedithio-TTF electron donor and its (1 : 1) triiodide radical cation salt; isomer effects in C–N⋯H–C interactions†
Abstract
A new isomer of the dissymmetric TTF derivative, 4-cyanobenzene-ethylenedithio-tetrathiafulvalene (4-CNB-EDT-TTF) (1), was prepared and compared with the previously reported 5-CNB-EDT-TTF. 1 was obtained in high yield, by a cross-coupling reaction with triethyl phosphite between 2-oxobenzo[d][1,3]dithiole-4-carbonitrile (Ia) and 5,6-dihydro-[1,3]dithiolo[4,5-b][1,4]dithiine-2-thione (Ib). The key compound for the synthesis of the ketone Ia was thione 2-thioxobenzo[d][1,3]dithiole-4-carbonitrile (Ia′), both compounds are reported. 1 as a new electron donor was characterized namely by cyclic voltammetry, NMR, UV-visible, IR and by single crystal X-ray diffraction. It crystallizes into two polymorphs (α-1 and β-1) with structures dominated by a large number of S-, C-, and N-mediated short contacts. One of the polymorphs (β-1) is present as a combination of C–N⋯H–C interactions associated with a head-to-head arrangement of donor molecules in paired layers (bilayers) described as weaker and stronger R23(10) synthons. A radical cation salt formulated as (4-CNB-EDT-TTF)I3 (2) was obtained by slow diffusion of solutions of the donor and iodine. In this salt, the (4-CNB-EDT-TTF)˙+ radical cations are associated into dyads with a strong pairing of the radical species in a singlet state. In 2, the donor dimers are connected to adjacent dimers through C–N⋯H–C interactions described as an R22(10) synthon. They present different sources of disorder on the carbon atoms of the dihydrodithiin ring, in the CN group position and on one of the triiodide anions. No relation between the disorders of the triiodide anions and the CN group positions was detected.


 Please wait while we load your content...
Please wait while we load your content...