A facile coordination precipitation route to prepare porous CuO microspheres with excellent photo-Fenton catalytic activity and electrochemical performance†
Abstract
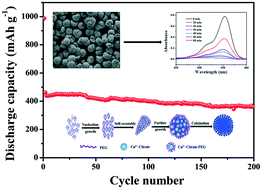
Porous CuO microspheres were prepared via a coordination precipitation route followed by a calcination process. The porous CuO microspheres have an average diameter of about 3–4 μm, and are composed of nanoparticles. The effect of the volume ratio of polyethylene glycol 400 and dimethylformamide, and the amount of citric acid on the morphology of the CuO products was investigated. The morphology and microstructures of the porous CuO microspheres can be controlled by optimizing the experimental conditions. The possible formation mechanism was proposed. As an anode material for lithium-ion batteries, the CuO microsphere electrode exhibits a high reversible discharge capacity of 623 mA h g−1 at 0.2 A g−1 and superior cycle stability with a high capacity retention of 77.9% of the initial discharge capacity (462 mA h g−1) after 200 cycles even though at a high current rate of 1 A g−1. The porous CuO microspheres can also serve as a potential photocatalyst for the degradation of three organic dyes including rhodamine B, methylene blue and methyl orange under visible light irradiation in the presence of hydrogen peroxide at ambient temperature. This excellent performance can be ascribed to the synergistic effect of porosity and hierarchical structure containing micro and nano features.


 Please wait while we load your content...
Please wait while we load your content...