Design, synthesis and characterization of copper-based coordination compounds with bidentate (N,N and N,O) ligands: reversible uptake of iodine, dye adsorption and assessment of their antibacterial properties†
Abstract
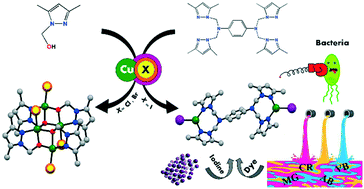
Four copper-based coordination compounds, namely [Cu2(tpmp)I2] (1) and [Cu2(tpmp)Cl2] (2) {tpmp = N,N,N′,N′-tetrakis(3,5-dimethyl-1-pyrazolyl)methyl-1,4-phenylenediamine} possess dinuclear structures, and [CuBr(LO)]4 (3) and [CuCl(LO)]4 (4) (LO− = 1-methoxy-3,5-dimethylpyrazole) with tetranuclear frameworks, were designed, synthesized and fully characterized by elemental analysis, IR, UV-vis, PXRD, TGA, CV and X-ray structural analysis. In the isostructures of 1 and 2 with an N,N donor ligand, the coordination environment around each of the copper atoms is a distorted trigonal planar with two imine nitrogen atoms of the two pyrazole rings from the tetradentate tpmp ligand and one terminal halogen atom. Compounds 3 and 4 with an N,O donor ligand have four and five coordination geometries around the copper atoms. In contrast to MOF materials, up to now few studies have been reported on simple and dinuclear complexes for adsorption processes. Therefore, in this work the iodine and dye adsorption behavior of these compounds was evaluated. Additionally, the uptake and release of iodine molecules were studied in detail for compounds 1 and 2 and the results confirmed their good performance. In contrast to 1 and 2, compounds 3 and 4 were inert with respect to the adsorption of iodine and dye molecules. The antibacterial activity of compounds 1–4 was also investigated.


 Please wait while we load your content...
Please wait while we load your content...