Controlling the size of Pt nanoparticles with a cationic surfactant, CnTABr†
Abstract
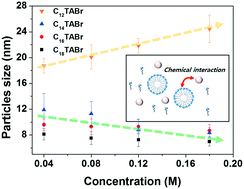
Aqueous-based colloidal synthesis using alkyltrimethylammonium bromide (CnTABr), a cationic surfactant, has attracted much attention recently as a simple and facile method to fabricate size- and shape-tunable metal nanoparticles. Despite the many related studies, however, the underlying synthesis mechanism and the exact role of the cationic surfactant have not yet been clarified. Here, we report how the size of Pt nanoparticles varies by using a wide range of chain lengths (n = 10, 12, 14, 16, and 18) and concentrations (0.005 to 0.18 M) of CnTABr in aqueous-based colloidal synthesis. Highly monodisperse Pt particles with a size between 8 and 26 nm were obtained and confirmed by TEM analysis. Significantly, unlike other surfactants, when CnTABr with a small chain length was used, the particle size increased with the concentration, which is different from the typical particle growth behavior by other surfactants. The UV-vis spectroscopy analysis results show that the chemical affinity between real precursors and micelles generated in solution is an important factor in determining the size of the Pt nanoparticles. Based on this insight, we successfully demonstrated that the particle size is also controllable by modifying the critical micelle concentration with a KBr additive. Our results provide useful guidelines for nanoparticle synthesis using cationic surfactants.


 Please wait while we load your content...
Please wait while we load your content...