Self-assembly of new M(ii) coordination polymers based on asymmetric 1,3,4-oxadiazole-containing ligands: effect of counterions and magnetic properties†
Abstract
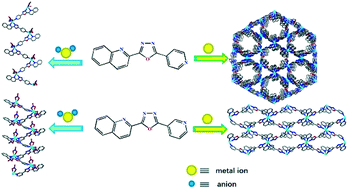
Two new unsymmetrical ligands, namely, 2-(pyridin-3-yl)-5-(quinolin-2-yl)-1,3,4-oxadiazole (L1) and 2-(pyridin-4-yl)-5-(quinolin-2-yl)-1,3,4-oxadiazole (L2), have been synthesized and their self-assembly reactions with MII(NO3)2 (M = Cd, Zn and Cu) and MII(ClO4)2 (M = Cd and Cu) have been investigated for the rational construction of porous coordination polymers. It was found that the counter-anion plays an important role in the structural assemblies. The strongly coordinating NO3− anions resulted in the formation of one-dimensional (1D) polymers (1–3 and 6). The weakly coordinating ClO4− anions gave rise to two-dimensional (2D) and three-dimensional (3D) porous coordination polymers (4 and 5). In polymers 1–3 and 6, the quinoline units play a critical role in the formation of a π–π stacking system, which further expands the dimensionality from one to three. Polymers 4 and 5 possess 1D rhombic and hexagonal channels, respectively. This study demonstrates that the asymmetric molecules L1 and L2 which contain monodentate and bidentate chelating binding sites can be very useful types of organic ligands to construct porous 2D- and 3D-cation frameworks. Although ab initio prediction of a particular superstructure from the starting materials and synthetic conditions is still impossible, some general principles are applicable for the observed structures and synthetic conditions. The photoluminescence of 1, 2, 4 and 6 and the magnetic properties of 3 and 5 are also studied.


 Please wait while we load your content...
Please wait while we load your content...