Unveiling the correlation between structural order–disorder character and photoluminescence emissions of NaNbO3†
Abstract
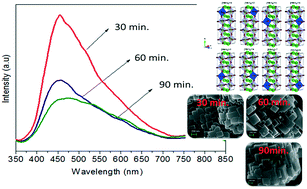
NaNbO3 samples were synthesized via a microwave-assisted hydrothermal method for different processing times (ranging from 30 to 90 minutes) at 180 °C using NaOH as a mineralizer. XRD and Rietveld refinement data confirmed the successful preparation of NaNbO3 with an orthorhombic (Pbma) crystallographic structure devoid of secondary phases. Raman scattering spectra reveal that the symmetry of the NaNbO3 space group has vibrational modes associated with the librations and internal vibrational modes of NbO6 the octahedron. The NaNbO3 particles grow in plate-like shape besides presenting high agglomeration degree. However, the particles assume parallelepiped-like shape as increasing synthesis time and the agglomeration degree becomes smaller. With an increase in the processing time, the optical band gap (3.24 eV (30 min), 3.32 eV (60 min), and 3.34 eV (90 min)) presents a slight difference in the density of intermediary energy levels within the band gap, which affects the electronic transitions. The broad band PL emission (350–800 nm) obtained at room temperature with a dominant PL sub-band at ∼455 nm (blue component) indicates that the changes in the electronic density are related to the defects of oxygen vacancies (in the bulk and surface) due to the structural disorder degree in the medium-range, i.e., local distortions of the NbO6 octahedron as result of changes in the Nb–O bond lengths and O–Nb–O bond angles, increasing the degree of structural disorder in the medium-range, and thereby a decline in the PL intensity with an increase in synthesis time. This medium-range structural disorder is able to transform the NaNbO3 ordered structure, which is constituted of 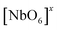 clusters, into an ordered-disordered NaNbO3 structure formed by
clusters, into an ordered-disordered NaNbO3 structure formed by 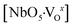 ,
, 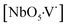 and
and 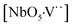 complex clusters.
complex clusters.


 Please wait while we load your content...
Please wait while we load your content...