Observing non-classical crystallisation processes in gypsum via infrared attenuated total reflectance spectroscopy†
Abstract
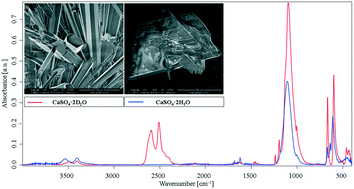
The nature of crystallisation processes is of major interest, as they are among the most frequently occurring reactions associated with a variety of relevant processes in chemistry, biochemistry, and geochemistry. In this study, an innovative approach towards fundamentally understanding crystallisation pathways in a seemingly simple system – gypsum – has been developed via infrared spectroscopic techniques. Specifically, infrared attenuated total reflection spectroscopy (IR-ATR) was instrumental in revealing detailed information on inter- and intramolecular interactions during gypsum crystallization via subtle changes in the vibrational spectra of the involved reactants. When applying D2O as an isotope marker, it was shown that isotopically labelled water may serve as a viable spectroscopic probe during mid-infrared (3–15 μm) studies providing unique insight into the crystallization process at molecular-level detail. In addition, it was revealed that H2O and D2O give rise to distinctly different reaction kinetics during the crystallization process.


 Please wait while we load your content...
Please wait while we load your content...