Solvent-mediated supramolecular templated assembly of a metal organophosphonate via a crystal–amorphous–crystal transformation†
Abstract
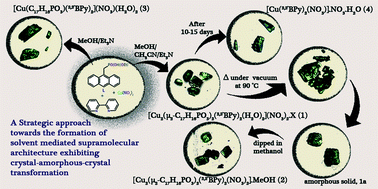
Three low-dimensional Cu(II)-based organophosphonates were synthesized: two molecular cationic complexes, [Cu2(μ2-L)2(2,2′BPy)2(H2O)2](NO3)2·S (1) and [Cu(L)(2,2′BPy)2]·NO3·(H2O)2 (3), and one neutral complex, [Cu2(μ2-L)2(2,2′BPy)2(NO3)2]·MeOH (2), assembled by the phosphonomonoester i.e., ethylhydrogen(anthracen-9-ylmethyl)phosphonate and an auxillary bidendate ligand (2,2′-bipyridine). Single-crystal X-ray diffraction analysis showed that complex 1 consisted of a dimer unit arranged supramolecularly with a nitrate anion and that these layers were further assembled via π⋯π interactions, which resulted in the formation of a porous supramolecular architecture. The cationic complexes 1 and 3 showed supramolecular templated assembly by simply altering the solvent, which resulted in a change in architecture. The remarkable change from a cationic complex to a neutral complex via the formation of an amorphous solid (1a) (a crystal–amorphous–crystal transformation) was observed when crystals of 1 were heated at 90 °C under vacuum. It is hypothesized that the uncoordinated nitrate ion moved from the lattice to the metal, resulting in the transformation of the cationic metal complex into a neutral metal complex. These complexes also showed luminescent behaviour.

 Please wait while we load your content...
Please wait while we load your content...