Can enantiomer ligands produce structurally distinct homochiral MOFs?†
Abstract
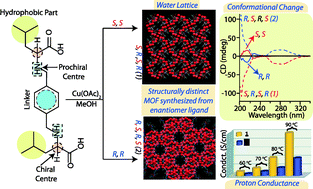
Here, we report a self-assembled homochiral metal–organic framework [Cu1.5(H2LL-leu)(Ac)H2O]n·3H2O (1) obtained from an L-leucine-derived ligand (H4LL-leu) and Cu(Ac)2·H2O in a 1 : 1 ratio. Coordination-induced conformational change in the ligand has been monitored by circular dichroism which has been further attested by synthesizing a D-leucine-containing enantiomer H4LD-leu and its Cu(II) complex [Cu1.5(H2LD-leu)H2O]n·10H2O (2). Structure determination revealed entirely different structures for the homochiral MOFs (1 and 2) obtained from the L/D-leucine-derived enantiomer ligands under analogous reaction conditions. Further, structural dissimilarity in these MOFs has been judicially supported by proton conductance studies. MOF 1 shows higher proton (10−5 S cm−1) conductance in comparison to 2 (10−6 S cm−1) due to dissimilar alignment of the hydrogen-bonded water molecules in the hydrophilic pocket as well as crystal packing.

 Please wait while we load your content...
Please wait while we load your content...