A facile strategy to adjust the density of planar triangle units in lead borate–nitrates†
Abstract
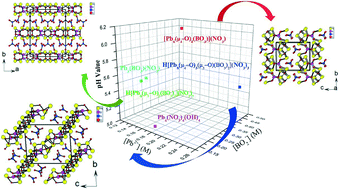
Three novel lead(II) borate–nitrates were obtained through a facile hydrothermal reaction by adjusting the concentrations of the starting materials, namely, [Pb6(μ4-O)4(BO3)](NO3) (1), H[Pb6(μ3-O)2(BO3)2](NO3)3 (2) and H[Pb8(μ4-O)3(μ3-O)(BO3)2](NO3)3 (3). All three compounds feature lead(II) oxo borate layers that are separated by nitrate anions. The 2D [Pb6(μ4-O)4(BO3)]+ layer parallel to the ab plane in 1 is built from 1D [Pb6(μ4-O)4]4+ chains along the a axis and bridging borate anions. The 2D H[Pb6(μ3-O)2(BO3)2]3+ layer in 2 which is perpendicular to the b axis is composed of “isolated” Pb2+ and tetranuclear [Pb4(μ3-O)2]4+ clusters interconnected by bridging BO3 groups. The [Pb8(μ4-O)3(μ3-O)(BO3)2]2+ (011) layer in 3 is composed of two types of lead(II) oxo chains, namely, 1D chains of [Pb4(μ4-O)2]4+ and 1D chains of [Pb4(μ4-O)(μ3-O)]4+, both elongated along the a axis, which are further interconnected by bridging borate anions. This study also demonstrates that a small change in the concentration of the starting materials could result in a product with a different density of the π-conjugated planar units.

 Please wait while we load your content...
Please wait while we load your content...