New 2D zinc phosphonates with heptanuclear units, reversible dehydration–hydration and bright luminescence†
Abstract
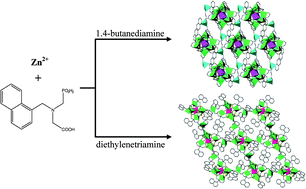
Two new zinc phosphonates with formulas [Zn18(L)12(H2N(CH2)4NH2)2]·25.5H2O (1) and [Zn9(L)6(HN(C2H4NH2)2)2]·10H2O (2) have been synthesized starting from zinc acetate, 1-C10H7-CH2N(CH2COOH)(CH2PO3H2) (H3L) and 1,4-butanediamine (for 1)/diethylenetriamine (for 2). Compounds 1 and 2 are characterized by single-crystal X-ray crystallography, powder XRD, elemental analysis, IR spectroscopy, and thermogravimetric (TG) analysis. In the crystal structures of compounds 1 and 2, Zn7 heptanuclear clusters are bridged by [Zn2(H2N(CH2)4NH2)] and [Zn(HN(C2H4NH2)2)] fragments, respectively, to form two-dimensional (2D) frameworks. Compound 1 is a dynamic framework upon the dehydration–hydration process and displays bright purple emission, while the framework of compound 2 is stable up to 250 °C under an air atmosphere. And the luminescence of compound 2 can be tuned by heating treatment.

 Please wait while we load your content...
Please wait while we load your content...