Energetic multi-component molecular solids of tetrafluoroterephthalic acid with some aza compounds by strong hydrogen bonds and weak intermolecular interactions of C–H⋯F and C–H⋯O†
Abstract
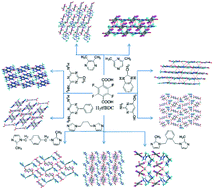
Tetrafluoroterephthalic acid, (H2tfBDC), forms nine novel crystals with a series of N-containing heterocycles: 2,3-dimethyl pyrazine (2,3-Pyr), 2,6-dimethyl pyrazine (2,6-Pyr), 2,4-diamino-6-methyl-1,3,5-triazine (dmt), Benzoguanamine (bga), 2-methylbenzimidazole (2-MeBzlmH), 1,4-bis(imidazol) butane (bimb), 2-amino-4-hydroxy-6-methyl pyrimidine (ahmp), 1,2-bis[(2-methylimidazol-1-yl)methyl] benzene (L7), and 1,4-bis[(2-methylimidazol-1-yl)methyl] benzene (L5). These crystal structures including salts/co-crystals/hydrates were analyzed and characterized by single crystal X-ray diffraction, IR, and TGA. Single crystal X-ray diffraction studies show that the huge numbers of hydrogen bonds play a significance part in assembling individual molecules into larger architectures, especially the strong N–H⋯O, O–H⋯O hydrogen bonds, and weak but highly directional C–H⋯O and C–H⋯F interactions exist commonly in all nine novel crystals. Crystal structure analysis shows that the F atom of the H2tfBDC participates in C–H⋯F hydrogen bond formation, producing different supramolecular synthons. More importantly, the N–H⋯O, O–H⋯O, and C–H⋯O hydrogen bonds are mainly involved in the supramolecular assembly of these molecules, but C–H⋯F hydrogen bonds convert two-dimensional networks into three dimensional, hence, the C–H⋯F interactions have a crucial role in the formation of higher-order supramolecular structures.

 Please wait while we load your content...
Please wait while we load your content...