Positional isomerism of unsymmetrical semirigid ligands toward the construction of discrete and infinite coordination architectures of zinc(ii) and cadmium(ii) complexes†
Abstract
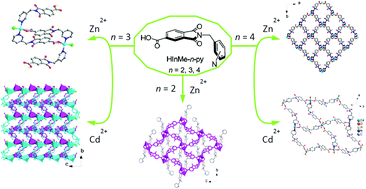
Three unsymmetrical semirigid ligands, HInMe-n-py (n = 2, 3, 4), showing positional isomerism have assembled with ZnCl2 and CdCl2 to yield supramolecular coordination assemblies. Compound [Zn(InMe-2-py)2]·1/2THF·1/2CH3OH·1/4H2O (1) has a two-dimensional rhombus gridlike (4,4) layer structure constructed from the dinuclear paddlewheel [Zn2(O2C)4N2] units with two pendant side-arms. Compound [Zn2Cl2(InMe-3-py)2(HInMe-3-py)2] (2) forms a discrete armed M2L2 macrocycle, which connects with others by intermolecular O–H⋯O and C–H⋯Cl hydrogen bonds to give rise to a decorated three-dimensional supramolecular pcu net. Compound [Zn(InMe-4-py)2]·1/2H2O (3) adopts a two-dimensional chiral wavy (4,4) square grid. Opposite-handedness chiral wavy grids are stacked in an ABAB type of sequence that has small voids accommodating lattice water molecules. Compound [CdCl(InMe-3-py)] (4) displays a two-dimensional achiral meso layer in which the opposite-handedness Cd–L helical chains are interconnected by the Cd–O bridging bonds and stitched by zigzag Cd–Cl chains. Compound [Cd(InMe-4-py)2(H2O)2] (5) features a two-dimensional rhombus gridlike (4,4)-net. Through the connections of net-to-net O–H⋯O hydrogen bonds, the grids are offset-stacked to be a hydrogen-bonded three-dimensional oblique pcu net. The structural diversity and complexity in these supramolecular coordination architectures are most likely attributed to the various coordination modes adopted by these N- and O-donor ligands of different substituent position and the coordination preference exhibited by the metal ions. Thermogravimetric (TG) analyses showed that all the frameworks of compounds 1–5 exhibit a thermal stability higher than 300 °C. In addition, the solid-state photoluminescent properties of compounds 1–5 were investigated.

 Please wait while we load your content...
Please wait while we load your content...