Two polymorphs and cocrystal of styryl-pyridine derivatives with tuned emission induced by Co2+ and Zn(phen)32+†
Abstract
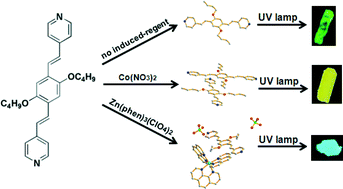
A styryl-pyridine derivative, 2,5-dibutoxy-1,4-bis[2-(4-pyridyl)ethenyl]benzene (L), has been designed and synthesised. Two polymorphs of L and crystals of [Zn(phen)3]2L(ClO4)4 were obtained by different crystallisation conditions. Polymorph-α was prepared by a solvent evaporation method. However, polymorph-β and a cocrystal of [Zn(phen)3]2L(ClO4)4 were obtained by different inducing reagents. It has been found that there are, in total, four independent conformations of the molecule L in the three kinds of crystals. We have calculated the standard molar enthalpy of formation (ΔHm) of each conformation and demonstrated that the stabilisation energy of each conformation significantly relies on both the planarity of the molecules (L) and bond length of conjugated bridges. Furthermore, the photophysical properties of the three kinds of crystals also exhibit differences in the vibration and absorption spectroscopies, solid-state photoluminescence and time-resolved fluorescence, which may arise from the chromophore's stacking modes and the intermolecular electronic interactions in the crystals. These results revealed that the optical properties of functional organic materials could be tuned by controlling their crystallisation environment for a specific molecule.

 Please wait while we load your content...
Please wait while we load your content...