Asymmetrical growth and dissolution along polar axis of α-resorcinol crystal: role of solvent and external environment†
Abstract
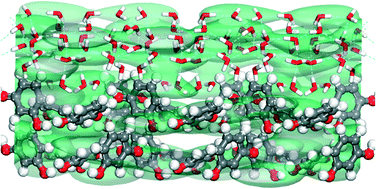
Asymmetrical growth and dissolution along the polar axis of α-resorcinol crystals from aqueous solution is an important and unresolved problem to date. Here we have computationally investigated the role of interfacial structure which occurs due to the ordering of solvent molecules at the crystal surface and external factors on the uneven growth and dissolution at the opposite and hemihedral faces of α-resorcinol crystals. Periodic dispersion corrected density functional theory has been employed to compute various energetics responsible for growth and dissolution. Our results show that not only the adsorption energy of the strongly adsorbed water molecules at the (011) face is higher than that at the (0![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif)
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) face but the adsorption energy of the rate-limiting resorcinol molecules at the former face is lower than that of the latter face. These energetics have been used to calculate the growth and dissolution rates of (0
) face but the adsorption energy of the rate-limiting resorcinol molecules at the former face is lower than that of the latter face. These energetics have been used to calculate the growth and dissolution rates of (0![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif)
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) and (011) faces from aqueous solution as a function of super(under)saturation at various temperatures which demonstrates the asymmetrical growth and dissolution along the polar axis. Our results show that the asymmetry in growth as well as dissolution along the polar axis of α-resorcinol crystals decreases with increasing temperature. Increasing supersaturation results in a decrease in growth asymmetry which is in good agreement with the experimental results available in the literature. On the other hand, our results predict that the dissolution asymmetry increases with increasing undersaturation.
) and (011) faces from aqueous solution as a function of super(under)saturation at various temperatures which demonstrates the asymmetrical growth and dissolution along the polar axis. Our results show that the asymmetry in growth as well as dissolution along the polar axis of α-resorcinol crystals decreases with increasing temperature. Increasing supersaturation results in a decrease in growth asymmetry which is in good agreement with the experimental results available in the literature. On the other hand, our results predict that the dissolution asymmetry increases with increasing undersaturation.

 Please wait while we load your content...
Please wait while we load your content...