One-shot versus stepwise gas–solid synthesis of iron trifluoride: investigation of pure molecular F2 fluorination of chloride precursors†
Abstract
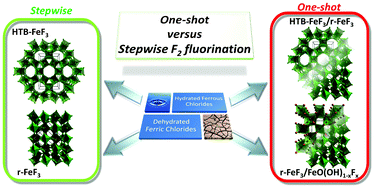
Iron trifluoride has been known for a long time and here we provide new insights into its detailed synthesis. Anhydrous iron trifluorides with different structures have been synthesized via one-shot and stepwise gas–solid fluorination under a gaseous flow of molecular fluorine from 150 °C. The degree of hydration of the precursors has a direct influence on the crystalline phase obtained: one-shot fluorination of FeCl2·4H2O leads to the hexagonal tungsten bronze-type FeF3 while fluorination of dehydrated iron chlorides gives the rhombohedral FeF3 with traces of oxy(hydroxy)fluorides. A stepwise procedure at 150, 250 and 350 °C tends to lead to pure phases and avoid the crystallization of oxy(hydroxy)fluorides in the final powder.

 Please wait while we load your content...
Please wait while we load your content...