Surfactant-free CuO mesocrystals with controllable dimensions: green ordered-aggregation-driven synthesis, formation mechanism and their photochemical performances†
Abstract
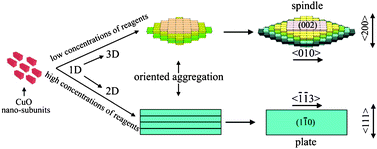
We have demonstrated the significant evidence on a green synthesis for the ordered-aggregation-driven growth from surfactant-free one-dimensional (1D) CuO nanosubunits into dimension-controlled mesostructures (three-dimensional (3D) mesospindles and two-dimensional (2D) mesoplates) by an additive-free complex–precursor solution route. Structural and morphological evolutions were investigated by X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), transmission electron microscopy (TEM), high-resolution transmission electron microscopy (HRTEM) and field-emission scanning electron microscopy (FESEM). The formation of CuO mesostructures here is essentially determined by the characteristic of [Cu(OH)4]2− precursors, and an oriented nanoparticle aggregation with tailoring shapes in different dimensions can be achieved in different concentration of reactants at higher reaction temperature. The 3D “layer-by-layer” growth of mesostructural CuO spindles is successfully synthesized in low concentrations of reagents, while the 2D “shoulder-by-shoulder” growth of mesostructural CuO plates is obtained in high concentrations of reagents. The study is of great importance in the bottom-up assembly of controllable ordering architectures, and offers a good opportunity to understand the fundamental significance for the investigation of the formation mechanism and growth process of surfactant-free CuO mesostructures with controllable aggregation-based behaviours. Additionally, we further demonstrated that such CuO mesocrystals could serve as a potential photocatalyst for the degradation of rhodamine B (RhB) under visible light irradiation in the presence of hydroxide water (H2O2). The results also suggest that these 3D mesostructural CuO spindles exhibit a higher adsorption and photocatalytic degradation of RhB than that of 2D mesostructural CuO plates.

 Please wait while we load your content...
Please wait while we load your content...