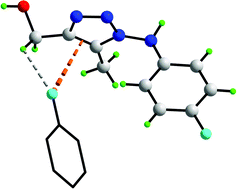
The presence of localised C–X⋯π [or C–X⋯π(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C)] interactions are shown to be pivotal in the crystal structures of (5-methyl-1-(4-X-arylamino)-1H-1,2,3-triazol-4-yl)methanol derivatives, X = H (1), F (2) and Cl (3). In the absence of halide (1), molecules aggregate into supramolecular chains via alternating ten-membered {⋯HOC2N}2 and 14-membered {⋯HN2C3O}2 synthons. Molecules assemble into a three-dimensional architecture via edge-to-face C–H⋯π(arene) interactions occurring between the phenyl rings. In the presence of halide (i.e. F (2) and Cl (3) in the 4-position of the phenyl ring), two-dimensional arrays are formed by interconnected ten-membered {⋯HOC2N}2 (as seen in 1) and 24-membered {⋯HO⋯NC2OH⋯N4H}2 hydrogen bonded synthons. The latter arrangement allows for the close approach of halide to the 1,2,3-triazole ring and the formation of C–X⋯π interactions which appear to be particularly significant in the case of Cl (3), as evidenced by systematic changes (i.e. elongation) in the geometric parameters within the five-membered ring. In this series of structures, the presence of C–X⋯π interactions is shown to moderate the supramolecular aggregation based on conventional hydrogen bonding.
C)] interactions are shown to be pivotal in the crystal structures of (5-methyl-1-(4-X-arylamino)-1H-1,2,3-triazol-4-yl)methanol derivatives, X = H (1), F (2) and Cl (3). In the absence of halide (1), molecules aggregate into supramolecular chains via alternating ten-membered {⋯HOC2N}2 and 14-membered {⋯HN2C3O}2 synthons. Molecules assemble into a three-dimensional architecture via edge-to-face C–H⋯π(arene) interactions occurring between the phenyl rings. In the presence of halide (i.e. F (2) and Cl (3) in the 4-position of the phenyl ring), two-dimensional arrays are formed by interconnected ten-membered {⋯HOC2N}2 (as seen in 1) and 24-membered {⋯HO⋯NC2OH⋯N4H}2 hydrogen bonded synthons. The latter arrangement allows for the close approach of halide to the 1,2,3-triazole ring and the formation of C–X⋯π interactions which appear to be particularly significant in the case of Cl (3), as evidenced by systematic changes (i.e. elongation) in the geometric parameters within the five-membered ring. In this series of structures, the presence of C–X⋯π interactions is shown to moderate the supramolecular aggregation based on conventional hydrogen bonding.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C)] interactions are shown to be pivotal in the crystal structures of (5-methyl-1-(4-X-arylamino)-1H-1,2,3-triazol-4-yl)methanol derivatives, X = H (1), F (2) and Cl (3). In the absence of
C)] interactions are shown to be pivotal in the crystal structures of (5-methyl-1-(4-X-arylamino)-1H-1,2,3-triazol-4-yl)methanol derivatives, X = H (1), F (2) and Cl (3). In the absence of 

 Please wait while we load your content...
Please wait while we load your content...