In our continuous efforts to explore the effects of metal ions, ligand structures, counter-anions on the structure and properties of metal–organic complexes, six new d10 metal coordination polymers with an anthracene-triazole ligand, 9,10-bis(triazol-1-ylmethyl)anthracene (L), {[Cu(L)I]}∞
(1), {[Ag(L)(BF4)]}∞
(2), {[Zn(L)(NO3)2]}∞
(3), {[Zn(L)(CH3OH)2(ClO4)2(H2O)]}∞
(4), {[Cd(L)(CH3OH)2(ClO4)2(H2O)]}∞
(5) and {[Hg(L)(Br2)]}∞
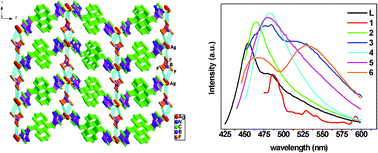
(6), were synthesized and structurally characterized by elemental analyses, IR spectroscopy and single-crystal X-ray diffraction. 1–3 and 6 have similar one-dimensional (1-D) chain structures in which 1, 3 and 6 were further linked into infinite two-dimensional (2-D) sheets by inter-chain C–H⋯I, π⋯π or C–H⋯Br weak interactions, respectively, while 2 was extended into three-dimensional (3-D) supramolecular network by Ag⋯F and Ag⋯N weak interactions. 4 and 5 possess similar (4,4) 2-D sheet structure containing only one type of rhombic grid, and the Zn/Cd atoms are regarded as the four-connected nodes and the ligand as bridges. Obviously, the structural differences among them are attributable to the different metal ions and counter anions. In 1–6, L adopts a trans-gauche conformation with the shortest N⋯N distance between the two N donors. Furthermore, the fluorescent properties of 1–6 and ligand L have been investigated.

 Please wait while we load your content...
Please wait while we load your content...