Small molecule binding to inhibitor of nuclear factor kappa-B kinase subunit beta in an ATP non-competitive manner†
Abstract
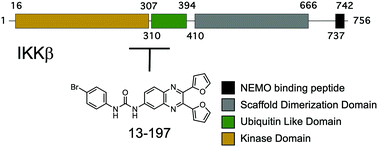
Inhibitor of nuclear factor kappa-B kinase subunit beta (IKKβ) is a key regulator of the cannonical NF-κB pathway. IKKβ has been validated as a drug target for pathological conditions, which include chronic inflammatory diseases and cancer. Pharmacological studies revealed that chronic administration of ATP-competitive IKKβ inhibitors resulted in unexpected toxicity. We previously reported the discovery of 13-197 as a non-toxic IKKβ inhibitor that reduced tumor growth. Here, we show that 13-197 inhibits IKKβ in a ATP non-competitive manner and an allosteric pocket at the interface of the kinase and ubiquitin like domains was identified as the potential binding site.


 Please wait while we load your content...
Please wait while we load your content...