Palladium catalyzed selective arylation of o-carboranes via B(4)–H activation: amide induced regioselectivity reversal†
Abstract
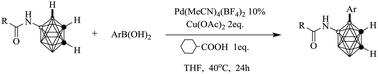
By changing the charge distribution of boron vertices via introducing an amide on cage B(9), the selective B(4) arylation of o-carboranes via Suzuki–Miyaura coupling has been developed. A series of o-carborane derivatives decorated with diverse active groups have been synthesized with moderate to good yields, which have been proved to be further transformed to a novel kind of tri-substituted nido-carborane fused oxazole with potential application in boron neutron capture therapy, organometallic as well as coordination chemistry.


 Please wait while we load your content...
Please wait while we load your content...