Highly efficient electrochemical ammonia synthesis via nitrogen reduction reactions on a VN nanowire array under ambient conditions†
Abstract
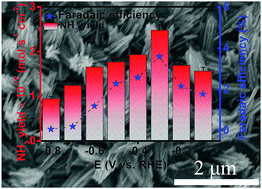
The development of a sustainable route to ammonia production is one of the most attractive targets in chemistry. The primary method of ammonia production, Haber–Bosch process, can bring about excessive consumption of fossil fuels and large CO2 emission. In this communication, we develop a VN nanowire array on carbon cloth (VN/CC) as a high-performance catalyst for the nitrogen reduction reaction (NRR) under ambient conditions. Such an electrocatalyst achieves high ammonia yield (2.48 × 10−10 mol−1 s−1 cm−2) and faradaic efficiency (3.58%) at −0.3 V versus RHE in 0.1 M HCl, outperforming most reported results for N2 fixation under ambient conditions, and even comparing favorably with those obtained under high temperatures and/or pressures. This work not only provides us an attractive catalyst material for the NRR in acidic media, but would also open up an exciting new avenue to the rational design and fabrication of transition metal nitrides for the NRR.


 Please wait while we load your content...
Please wait while we load your content...