Anion binding modes in cis–trans-isomers of a binding site–fluorophore–π-extended system†
Abstract
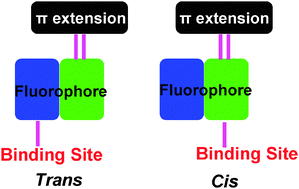
The cis- and trans-configurational isomers of 3 are designed and investigated as chemodosimeters for CN− ions for the first time. The cis-3 reveals a NIR absorbance at 717 nm (vs. trans-3, 620 nm), and such a configurational difference can be attributed to the acidity as well as position of the binding site (CH group). A probable sensing mechanism involving cyanide-driven carbanion electron-transfer oxidation is proposed.

 Please wait while we load your content...
Please wait while we load your content...