Unexpected formation of a cyclic vinylene sulfate in the synthesis of ethynyl-substituted acenes†
Abstract
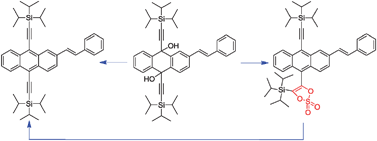
(E)-2-Styrylanthracene derivatives containing triisopropylsilylacetylene groups at the 9 and 10 positions were synthesized and characterized. The electronic properties have been studied by DFT calculations,

 Please wait while we load your content...
Please wait while we load your content...