Brimonidine-LAPONITE® intravitreal formulation has an ocular hypotensive and neuroprotective effect throughout 6 months of follow-up in a glaucoma animal model†
Abstract
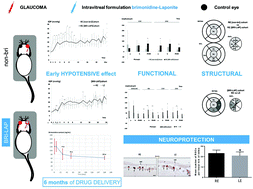
Intravitreal administration is widely used in ophthalmological practice to maintain therapeutic drug levels near the neuroretina and because drug delivery systems are necessary to avoid reinjections and sight-threatening side effects. However, currently there is no intravitreal treatment for glaucoma. The brimonidine-LAPONITE® formulation was created with the aim of treating glaucoma for extended periods with a single intravitreal injection. Glaucoma was induced by producing ocular hypertension in two rat cohorts: [BRI-LAP] and [non-bri], with and without treatment, respectively. Eyes treated with brimonidine-LAPONITE® showed lower ocular pressure levels up to week 8 (p < 0.001), functional neuroprotection explored by scotopic and photopic negative response electroretinography (p = 0.042), and structural protection of the retina, retinal nerve fibre layer and ganglion cell layer (p = 0.038), especially on the superior-inferior axis explored by optical coherence tomography, which was corroborated by a higher retinal ganglion cell count (p = 0.040) using immunohistochemistry (Brn3a antibody) up to the end of the study (week 24). Furthermore, delayed neuroprotection was detected in the contralateral eye. Brimonidine was detected in treated rat eyes for up to 6 months. Brimonidine-LAPONITE® seems to be a potential sustained-delivery intravitreal drug for glaucoma treatment.


 Please wait while we load your content...
Please wait while we load your content...