The promising indicators of the thermal and mechanical properties of collagen from bass and tilapia: synergistic effects of hydroxyproline and cysteine†
Abstract
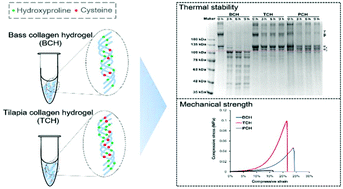
Collagen has been widely documented as one of the most promising and competitive biomaterials for tissue engineering and medical applications. However, the properties of collagen differ from one source to another. In the present study, type I collagen (COL-I) was extracted and purified from the skins of Japanese sea bass (Lateolabrax japonicus) and Nile tilapia (Oreochromis niloticus). Ultraviolet (UV) spectroscopy, Fourier transform infrared spectroscopy (FTIR) and SDS-PAGE were performed to characterize both COL-Is. The denaturing temperature of bass collagen (BC) was observed to be 27.2 °C, and 35.3 °C for tilapia collagen (TC). The content of hydroxyproline was 13.4% in TC, which was similar to that in porcine collagen (PC, 13.6%) and higher than that in BC (10.3%), while the content of cysteine in TC (0.87%) was significantly higher than that in PC (0.04%) and BC (0.35%). After incubation at different temperatures for 9 h, more degraded collagen bands appeared in the BC hydrogel (BCH) group than in the TC hydrogel (TCH) group, indicating that TCH exhibited better thermal stability than BCH. The thermal stabilities of TCH and PC hydrogel (PCH) were similar. The compressive stress of TCH was up to 0.099 MPa, while it was 0.047 MPa for BCH and 0.003 MPa for PCH. These results demonstrated that the content of amino acids (especially hydroxyproline and cysteine) has a synergistic effect on the thermal and mechanical properties of BCH, TCH and PCH, which would be an indicator of the thermal and mechanical properties of collagen hydrogels in future studies.


 Please wait while we load your content...
Please wait while we load your content...