Chemical characterization of different gourmet table salts by atomic spectrometry and FT-Raman spectroscopy†
Abstract
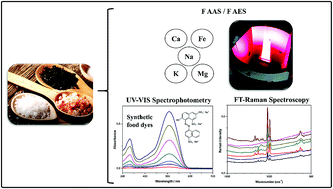
The contents of Ca, Fe, K, Mg and Na were studied in 23 common and gourmet salt samples by atomic spectrometry after an acid dissolution procedure using diluted nitric acid. Under optimized conditions, no matrix effects were observed when aqueous calibration was compared to standard addition methods. The method's accuracy was evaluated by analyte recovery experiments (recoveries ranging from 94 to 109%) and the limits of detection and quantification varied from 1.0 to 22 mg kg−1 and from 3.3 to 76 mg kg−1, respectively, which are adequate for the determination of essential mineral elements in these kinds of samples. To interpret the results, principal component analysis was used and showed that some Himalayan salts had similar Ca and Mg contents among different samples. On the other hand, blue Persian salts and low-sodium salts had the highest K concentration levels. In addition, the Hawaiian black salt samples had similar Na and Fe concentrations among different samples. In relation to the concentration of analytes, the results suggest that these types of salts cannot be considered to be a source of these minerals (Ca, Fe, and Mg), contrary to widespread statements in popular media. With respect to the Himalayan samples' authenticity, both UV-VIS spectrophotometry and FT-Raman spectroscopy were used to investigate the presence of ordinary colorants E122 and E124 in commercial samples, and no evidence of adulteration or fraud was observed.


 Please wait while we load your content...
Please wait while we load your content...