Dispersion solid-phase extraction of flavonoid with amphiphilic monomers N-vinyl pyrrolidone and 1H,1H,7H-dodecafluoroheptyl methacrylate based poly(styrene-divinylbenzene) and silica†
Abstract
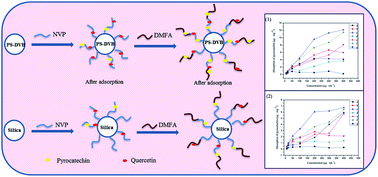
A range of materials were synthesized based on different matrices of poly(styrene-divinylbenzene) (PS-DVB) and silica (Si). N-vinyl pyrrolidone (NVP) and 1H,1H,7H-dodecafluoroheptyl methacrylate (DMFA) were the functional monomers for the synthesis of PS-DVB–NVP, PS-DVB–NVP–DMFA, Si–NVP and Si–NVP–DMFA. The structures of the obtained materials were characterized by scanning electron microscopy, Fourier transform infrared spectroscopy, and Brunauer–Emmett–Teller and thermogravimetric analysis. The results demonstrated that NVP and DMFA can change the structure of the matrix pores. Using response surface methodology (RSM), the optimal proportion for synthesizing the materials was 330.00 μL of DMFA and 11.00 mg of AIBN at 80 °C. P–N–F showed the best recognition for pyrocatechin and quercetin, and the adsorption abilities were 12.14 μg mg−1 and 6.76 μg mg−1, respectively. In terms of applications, the method's recoveries of pyrocatechin and quercetin were in the range of 78.06–83.43% and 79.05–83.63% with associated intra-day relative standard deviations of less than 2.92% and 2.84%, respectively. P–N–F showed satisfactory results in that recoveries for pyrocatechin and quercetin were 83.43% and 83.63%, respectively. This showed potential for the rapid purification of flavonoids in an apple sample combined with dispersion solid-phase extraction (DSPE).


 Please wait while we load your content...
Please wait while we load your content...