Highly selective and sensitive optical probe for Fe3+ based on a water-soluble squarylium dye†
Abstract
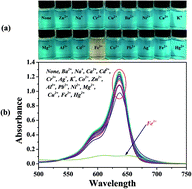
A highly selective and sensitive water-soluble optical probe based on a squarylium dye, 2,3,3-trimethyl-3H-indolenine-5-sulfonate squaraine (SQ) has been successfully synthesized. The as-prepared SQ can efficiently recognize iron(III) ions in the presence of competing cations (Na+, Mg2+, Al3+, Cr3+, Zn2+, K+, Ca2+, Ba2+, Pb2+, Ni2+, Co2+, Ag+, Cu2+, Cd2+) in deionized water. After Fe3+ was added, the absorbance and fluorescence of the SQ water solution changed, and the color changed from sky blue to pale yellow. The binding constant (Ka) of SQ–Fe3+ was calculated to be about 1.14 × 104 M−1. Correspondingly, the chelating mode of SQ–Fe3+ was confirmed by Job's plot, Fourier transform infrared (FT-IR) and 1H nuclear magnetic resonance (1H NMR). “Naked eye” detection was performed, which indicated the potential application of SQ for convenient, selective and fast detection of Fe3+.


 Please wait while we load your content...
Please wait while we load your content...