Development and validation of an LC-MS/MS method for simultaneous quantification of hesperidin and hesperetin in rat plasma for pharmacokinetic studies†
Abstract
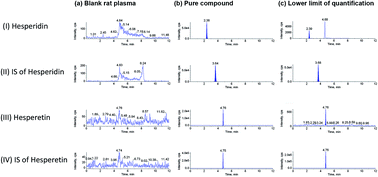
A simple and sensitive method for the determination of hesperidin and hesperetin was necessary for a pharmacokinetic (PK) study in Wistar rats. Polarity-switching mode was utilized to acquire positive ion electrospray data for hesperidin and neohesperidin dihydrochalcone-d3, and negative ionization data for hesperetin and rac-hesperetin-d3. The analytes were investigated in the multiple reaction monitoring mode as the precursor/product ion pair of m/z 611.2 → 303.1 for hesperidin, m/z 616.3 → 308.3 for neohesperidin dihydrochalcone-d3, m/z 301.3 → 164.1 for hesperetin, and m/z 304.3 → 164.1 for rac-hesperetin-d3. Hesperidin and hesperetin in rat plasma were separated with solid-phase extraction using a HyperSep Retain PEP column and separated on a Hypersil GOLD Phenyl reversed-phase column using a gradient elution of 0.1% formic acid in water and acetonitrile. Hesperidin was validated over the concentration range of 1–1000 ng mL−1, and hesperetin was validated over the range of 0.2–100 ng mL−1, both with r > 0.999. The respective within- and between-run precisions were 2.06–9.54% and 2.11–7.76%, and the within- and between-run accuracies were −6.52% to 3.82% and −1.62% to 2.33%, respectively, for both analytes. Extraction recoveries of these analytes were >87%. The matrix effects were between 94.7% and 113.6%. All of the analytes were stable during the assay and storage in Wistar rat plasma. The lower limit of quantification of the active metabolite hesperetin is up to 200 pg mL−1, so complete plasma concentration–time profiles can be obtained by the LC-MS/MS method without the need for enzymatic hydrolysis of the serum sample.


 Please wait while we load your content...
Please wait while we load your content...