Pharmacokinetics and brain distribution studies of ginsenoside Rd in rats via intranasal administration by LC-MS/MS
Abstract
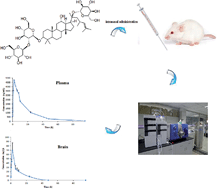
Ginsenoside Rd was shown to have protective effects against several injuries and efficient for the treatment of acute ischemic stroke. Some research studies of ginsenoside Rd in the past mainly focused on the pharmacokinetics after intravenous and oral administration. However, we still lack some basic knowledge about the plasma pharmacokinetics and brain distribution of ginsenoside Rd by any other route, such as intranasal administration. It was found that intranasal administration exhibited good brain-targeting. In this study, a sensitive LC-MS/MS method was developed and validated for the determination of ginsenoside Rd in rat plasma and brain tissue. Detection was performed on an ACQUITY UPLC™ BEH C18 column using gradient elution with a flow rate of 0.2 mL min−1. Mass spectrometry was operated in selected reaction monitoring mode using a negative electrospray ionization interface. The method was linear over the concentration range of 1.0–1000 ng mL−1, and the lower limit of quantification was 1.0 ng mL−1 for ginsenoside Rd. The method was validated in terms of specificity, linearity, intra- and inter-day precision (<12.39%), accuracy (within ±10.1%), dilution integrity, recovery, matrix effects and stability, and has been successfully applied to the pharmacokinetic study of ginsenoside Rd in rats after intranasal administration and evaluation of the brain targeting of ginsenoside Rd.

 Please wait while we load your content...
Please wait while we load your content...