Determination of α-hederin in rat plasma using liquid chromatography electrospray ionization tandem mass spectrometry (LC-ESI-MS/MS) and its application to a pharmacokinetic study
Abstract
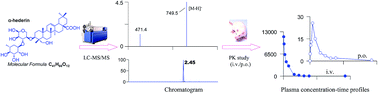
The evaluation of efficacy and safety should be paralleled with the assessment of comprehensive pharmacokinetic (PK) properties for a drug candidate, and a robust bioanalytical method is a prerequisite for obtaining PK information. α-Hederin is reported to have various in vitro and in vivo activities; however, very little is known about its PK and metabolic characteristics. In this study, we have developed an efficient LC-ESI(−)-MS/MS assay for α-hederin and its sapogenin hederagenin in rat plasma. Sample cleanup involved methanol precipitation for identification analysis and liquid–liquid extraction with ethyl acetate for quantification assay. LC analysis was performed under reversed-phase conditions in the modified “pulse gradient elution” mode. Analyte identification and quantification were conducted using multiple reaction monitoring (MRM) mode with euscaphic acid as the internal standard. Under these conditions, deglycosylated metabolites and their sulfate conjugates were detected; however, hederagenin was not detected in rat plasma samples after both oral and intravenous treatments. The mean plasma clearance (CL), volume of distribution (VSS) and elimination half-life (t1/2) of α-hederin were 0.24 L h−1 kg−1, 0.25 L kg−1 and 2.67 h, respectively. The oral bioavailability (F) of α-hederin in rats was about 0.14%, which might result from the poor intestinal absorption and/or extensive biliary excretion. It is hoped that this validated method will be useful for future PK studies of α-hederin.

 Please wait while we load your content...
Please wait while we load your content...