Prussian blue nanoparticles: a simple and fast optical sensor for colorimetric detection of hydralazine in pharmaceutical samples
Abstract
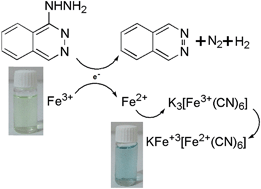
A simple, inexpensive and fast colorimetric method was developed for the detection of hydralazine, which is an antihypertensive drug. The method is based on the reaction of HY with ferric ions (as oxidizing agent) in the presence of ferricyanide ion and formation of prussian blue nanoparticles (KFeIII[FeII(CN)6]). A UV-vis spectrophotometer was used to monitor the changes of the absorption intensity of the prussian blue nanoparticles. These nanoparticles exhibited a strong UV-vis extinction band at 700 nm. Change in color of the solution, which is directly related to the HY concentration, could be easily observed with the naked eye in the presence of a sub-ppm level of HY. The effect of several reaction variables on the rate of the PBNP formation was studied and optimized. A linear relationship between absorbance intensity of PBNPs and the concentration of HY over a range of 0.4 μg mL−1 to 2.0 μg mL−1 with correlation coefficient (R2) of 0.9967 was observed; moreover, the detection limit was found to be 0.33 μg mL−1. The proposed method showed a good detection limit, and compared to other methods it is very fast, simple and inexpensive. Furthermore, the proposed method was successfully applied for the determination of HY concentration in pharmaceutical samples with satisfactory results.

 Please wait while we load your content...
Please wait while we load your content...