Towards an ideal method for analysis of lisinopril in pharmaceutical formulations using a tris(2,2′-bipyridyl)-ruthenium(ii)-peroxydisulfate chemiluminescence system in a two chip device
Abstract
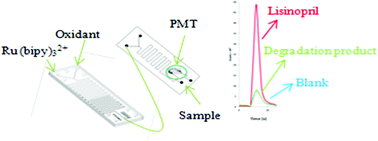
A novel, rapid, selective and sensitive method has been developed for analysis of lisinopril (LIS) in pharmaceutical formulations using a tris(2,2′-bipyridyl)-ruthenium(II) (Ru(bipy)32+) peroxydisulfate chemiluminescence (CL) system in a two chip device. The parameters that influence the CL signal intensity were carefully optimized. These include pH, flow rates and concentration of reagents used. Under optimum conditions, a linear calibration curve between 0.25 and 50.0 μg mL−1 was obtained. About 180 samples were analyzed per hour while the detection limit was found to be 0.067 μg mL−1. The method was applied for analysis of LIS in pharmaceutical products and was found to be free from interferences of acid-induced degradation (AID) products and other ingredients usually present in these preparations.
 Please wait while we load your content...
Please wait while we load your content...