Biocompatible Ag2S quantum dots for highly sensitive detection of copper ions†
Abstract
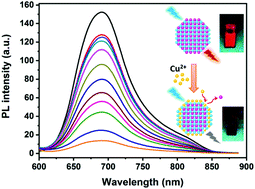
A simple aqueous approach for synthesizing biocompatible red and near-infrared (NIR) emitting Ag2S quantum dots (QDs) at low temperature without a required oxygen-free process has been developed. The fluorescence of the obtained red emitting Ag2S QDs could be selectively quenched by Cu2+. Based on this, a novel Ag2S QD based fluorescent sensor for highly selective and sensitive detection of Cu2+ was developed. This method showed a low limit of detection (LOD) of 27.6 nM for Cu2+ sensing in a wide linear range concentration of 25 nM–10 μM. The quenching mechanism was discussed by the time-resolved photoluminescence, absorption spectra, TEM and energy-dispersive X-ray (EDX) results. Cation exchange of Ag2S QDs between Cu(II) and Ag(I), and dynamic quenching due to excited state electron transfer from Ag2S QDs to Cu2+ were considered to be the quenching mechanism. Furthermore, to understand the interaction between Ag2S QDs and Cu2+, the thermodynamic process was investigated by means of isothermal titration calorimetry (ITC). The values of ΔH, ΔS and ΔG were calculated from the ITC results to be −79.43 kJ mol−1, −142.18 J K−1 mol−1 and −37.06 kJ mol−1, respectively. The Ag2S QD based Cu2+ detection had advantages of nontoxicity, fast response, high sensitivity and selectivity, wide linear range and easy preparation.


 Please wait while we load your content...
Please wait while we load your content...