Voltammetric determination of diffusion coefficients in polymer membranes†
Abstract
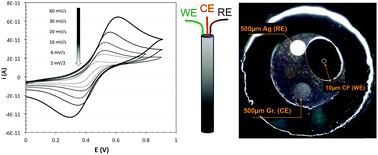
The diffusion-controlled transport of ions and molecules through polymer membranes utilized in chemical and biosensors is often the key factor determining the response characteristics of these sensors. In this paper, a simple voltammetric method is described for the determination of diffusion coefficients of redox molecules in resistive polymer membranes using a planar electrochemical cell (PEC) with a 5 μm radius carbon fiber as working electrode. In the proposed method, the diffusion coefficients are assessed from the scan rate dependence of the peak (or limiting) currents in linear sweep or cyclic voltammograms. The accuracy of the method is estimated through simulations using the method of Nicholson and Shain describing the quantitative relationship between the measured peak current (ip) and the square root of the scan rate (v1/2). The proposed method has been used for measuring the diffusion coefficients of ferrocene derivatives in highly resistive aqueous solutions, organic solvents, plasticizers, and plasticized PVC membranes. The measured diffusion coefficients are in agreement with theoretical models and previously reported values.


 Please wait while we load your content...
Please wait while we load your content...