Electroanalysis of pM-levels of urokinase plasminogen activator in serum by phosphorothioated RNA aptamer†
Abstract
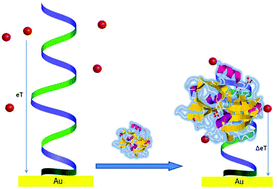
Protein biomarkers of cancer allow a dramatic improvement in cancer diagnostics as compared to the traditional histological characterisation of tumours by enabling a non-invasive analysis of cancer development and treatment. Here, an electrochemical label-free assay for urokinase plasminogen activator (uPA), a universal biomarker of several cancers, has been developed based on the recently selected uPA-specific fluorinated RNA aptamer, tethered to a gold electrode via a phosphorothioated dA tag, and soluble redox indicators. The binding properties of the uPA–aptamer couple and interference from the non-specific adsorption of bovine serum albumin (BSA) were modulated by the electrode surface charge. A nM uPA electroanalysis at positively charged surfaces, complicated by the competitive adsorption of BSA, was tuned to the pM uPA analysis at negative surface charges of the electrode, being improved in the presence of negatively charged BSA. The aptamer affinity for uPA displayed via the binding/dissociation constant relationship correspondingly increased, ca. three orders of magnitude, from 0.441 to 367. Under optimal conditions, the aptasensor allowed 10−12–10−9 M uPA analysis, also in serum, being practically useful for clinical applications. The proposed strategy for optimization of the electrochemical protein sensing is of particular importance for the assessment and optimization of in vivo protein ligand binding by surface-tethered aptamers.
- This article is part of the themed collection: In vivo analysis

 Please wait while we load your content...
Please wait while we load your content...