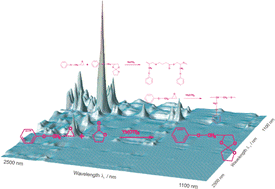
The spiroorthoester synthesis includes several competitives reactions. A way of determining the reactions that are taking place and their sequential order, is presented. The reaction between the phenylglycidylether and γ-butyrolactone to obtain a spiroorthoester has been monitored by near-infrared spectroscopy (NIR). In addition to the formation of the corresponding spiroorthoester, some parallel processes can occur. By means of two-dimensional correlation analysis, only one reaction is postulated, the one corresponding to the spiroorthoester formation. This was confirmed by recording the NMR spectra of the final product. Applying multivariate curve resolution-alternating least squares (MCR-ALS) to the NIR spectra obtained during the reaction, it has been possible to obtain the concentration values of the species involved in the reaction. The recovered spectra were compared with the experimentally recorded spectra for the reagents (phenylglycidylether, γ-butyrolactone) and the final product (spiroorthoester) and the correlation coefficients were, in all cases, higher than 0.990. The maximum and minimum limits associated with the ALS solutions were calculated, making it possible to limit to a considerable extent the ambiguity that is characteristic of these curve resolution methods.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...