Hydrogen storage capacity, strain-improved formation enthalpy, desorption temperature, and high energy harvesting performance of SrGaH5
Abstract
The study of hydrogen storage (HS) characteristics in complex hydrides has become a compelling goal in recent times. Therefore, we explore the SrGaH5 hydride by focusing on its potential for HS utilization and various physical aspects via first-principles calculations under hydrostatic strain. The results indicate that strain has a positive impact on the system’s thermodynamics, which enhances its potential for practical applications as a HS material. The formation enthalpy and desorption temperature decrease to −42.93/−45.18 kJ per mol H2 and 306.67/322.74 K under −5%/+5% strain from the unstrained values of −48.03 kJ per mol H2 and 343.13 K, respectively. These values are nearly ideal, approaching the theoretical values of −40 kJ per mol H2 and 233–333 K, making it a promising candidate for HS uses. Furthermore, the volumetric hydrogen capacity is enhanced to 17% at −5% and the system exhibits a high gravimetric hydrogen capacity of 3.10%, which is good enough for practical purposes. Along with this, the system is found to be mechanically stable, where the calculated 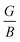 ratio and Poisson’s ratio further imply the existence of ionic bonding character in the material. Further, the non-negative phonon frequency curves validate the dynamical stability of the structure. The electronic structure calculations predicted an insulating behavior with an indirect energy gap of 3.58–2.43 eV for the considered strain range. Surprisingly, the thermoelectric performance of the system has been significantly improved under tensile strain as the figure of merit reaches a high value of 0.61 at 550 K under +5% strain. This is because the mean free path of phonons is reduced as the temperature rises due to an increase in phonon scattering driven by greater lattice vibrations, which causes a decrease in lattice thermal conductivity. Hence, the hydrogen storage capabilities and other aspects of the material indicate that it is an appropriate candidate for HS and might be crucial in various energy-generating applications.
ratio and Poisson’s ratio further imply the existence of ionic bonding character in the material. Further, the non-negative phonon frequency curves validate the dynamical stability of the structure. The electronic structure calculations predicted an insulating behavior with an indirect energy gap of 3.58–2.43 eV for the considered strain range. Surprisingly, the thermoelectric performance of the system has been significantly improved under tensile strain as the figure of merit reaches a high value of 0.61 at 550 K under +5% strain. This is because the mean free path of phonons is reduced as the temperature rises due to an increase in phonon scattering driven by greater lattice vibrations, which causes a decrease in lattice thermal conductivity. Hence, the hydrogen storage capabilities and other aspects of the material indicate that it is an appropriate candidate for HS and might be crucial in various energy-generating applications.



 Please wait while we load your content...
Please wait while we load your content...