Assessment of the interaction between sodium dodecyl sulfate and trypsin enzyme through micellization and thermodynamic analysis
Abstract
This study investigates the micellization behavior of a system comprising sodium dodecyl sulfate (SDS) and a biologically relevant protein, trypsin as well as the effects of different sodium salts and temperature on that system. The results reveal complex interactions among the surfactants, enzymes, and electrolytes, providing insights into the physicochemical mechanisms governing protein–surfactant systems. By systematically varying the concentrations of sodium salts (NaBr, CH3COONa(NaOAc), Na2SO4, and Na3PO4) and modulating the temperature from 290.55 K to 320.55 K, this research explores the self-association of SDS in the presence of trypsin. Conductometric analysis shows that sodium electrolytes reduce the critical micelle concentration (CMC) values of SDS, thereby enhancing micellization. Temperature influences this effect by generally favoring the micelle formation at lower temperatures while destabilizing the micelles at higher temperatures. The Gibbs free energy change 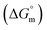 was found to be consistently negative (from −31.40 to −34.88 kJ mol−1), indicating the spontaneity of the micellization. Temperature-dependent variations in standard enthalpy
was found to be consistently negative (from −31.40 to −34.88 kJ mol−1), indicating the spontaneity of the micellization. Temperature-dependent variations in standard enthalpy 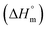 and entropy
and entropy 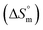 reveal that the entropy changes control the micelle formation at lower temperatures, while at higher temperatures, both entropy and enthalpy jointly control the process. Further, a molecular docking study was carried out using the CB-Dock2 online docking server with AutoDock Vina, which confirmed the binding interactions between the SDS surfactant and the trypsin protein. These findings have implications for biochemical processes, pharmaceutical formulation, and enzyme-based applications, where protein–surfactant interactions are critical.
reveal that the entropy changes control the micelle formation at lower temperatures, while at higher temperatures, both entropy and enthalpy jointly control the process. Further, a molecular docking study was carried out using the CB-Dock2 online docking server with AutoDock Vina, which confirmed the binding interactions between the SDS surfactant and the trypsin protein. These findings have implications for biochemical processes, pharmaceutical formulation, and enzyme-based applications, where protein–surfactant interactions are critical.



 Please wait while we load your content...
Please wait while we load your content...