Catalytic investigation of hyaluronic acid-stabilized Ag nanoparticles as non-toxic nanocatalysts in the oxidation of morin†
Abstract
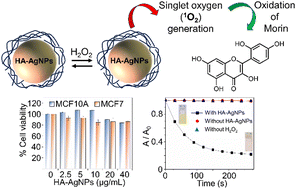
Catalytic oxidation is a widely used technology to eliminate undesired organic substances from water resources. Transition metal catalysts have a critical role to play in catalytic advanced oxidation; however, the toxicity of these catalysts is a major threat to human health and the environment. Therefore, research on the development of efficient oxidation catalysts with reduced toxicity is extremely critical. Herein, we report the catalytic investigation of hyaluronic acid-functionalized silver nanoparticles (HA-AgNPs) as an efficient and non-toxic catalyst in the degradation of morin dye as a model compound. The as-synthesized nanoparticles are characterized by different analytical methods such as ultraviolet-visible (UV-vis) spectroscopy, scanning transmission electron microscopy (STEM), X-ray diffraction (XRD), dynamic light scattering (DLS) and zeta potential. The particle size and charge of the HA-AgNPs are found to be 23.9 ± 8.3 nm and −43.5 ± 0.8 mV, respectively. The catalytic activity of HA-AgNPs has been assessed in the oxidation of morin with H2O2. The catalytic studies reveal that the oxidation follows first order reaction kinetics with an apparent rate constant of 1.01 × 10−2 s−1 and the degradation of morin has been completed within 5 min, indicating outstanding catalytic properties of HA-AgNPs. The cytotoxicity of HA-AgNPs was further evaluated by MTT assay and the results show that these nanoparticles are non-toxic to MCF-10A non-tumorigenic breast epithelial cells and MCF-7 breast cancer cells.


 Please wait while we load your content...
Please wait while we load your content...