The sliding motility of the bacilliform virions of Influenza A viruses†
Abstract
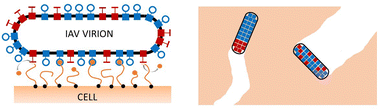
Influenza A virus (IAV) infection relies on the action of the hemagglutinin (HA) and neuraminidase (NA) membrane proteins. The HA ligands anchor the IAV virion to the cell's surface by binding the sialic acid (SA) present on the host's receptors while NA is an enzyme capable of cleaving the SA from the extracellular environment. It is believed that the activity of NA ligands increases the motility of the virions favoring the propagation of the infection. In this work, we develop a numerical framework to study the dynamics of a virion moving across the cell surface for timescales much bigger than the typical ligand–receptor reaction times. We find that the rates controlling the ligand–receptor reactions and the maximal distance at which a pair of ligand–receptor molecules can interact greatly affect the motility of the virions. We also report on how different ways of organizing the two types of ligands on the virions' surface result in different types of motion that we rationalize using general principles. In particular, we show how the emerging motility of the virion is less sensitive to the rate controlling the enzymatic activity when NA ligands are clustered.


 Please wait while we load your content...
Please wait while we load your content...