Coupling of α-bromoamides and unactivated alkenes to form γ-lactams through EDA and photocatalysis†
Abstract
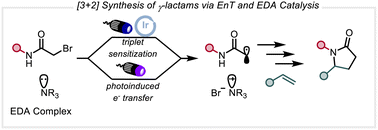
γ-Lactams are prevalent in small-molecule pharmaceuticals and provide useful precursors to highly substituted pyrrolidines. Despite numerous methods for the synthesis of this valuable motif, previous redox approaches to γ-lactam synthesis from α-haloamides and olefins require additional electron withdrawing functionality as well as N-aryl substitution to promote electrophilicity of the intermediate radical and prevent competitive O-nucleophilicity about the amide. Using α-bromo imides and α-olefins, our strategy enables the synthesis of monosubstituted protected γ-lactams in a formal [3 + 2] fashion. These species are poised for further derivatization into more complex heterocyclic scaffolds, complementing existing methods. C–Br bond scission occurs through two complementary approaches, the formation of an electron donor–acceptor complex between the bromoimide and a nitrogenous base which undergoes photoinduced electron transfer, or triplet sensitization with photocatalyst, to furnish an electrophilic carbon-centered radical. The addition of Lewis acids allows for further increased electrophilicity of the intermediate carbon-centered radical, enabling tertiary substituted α-Br-imides to be used as coupling partners as well as internal olefins.


 Please wait while we load your content...
Please wait while we load your content...