The thiol-sulfoxonium ylide photo-click reaction for bioconjugation†
Abstract
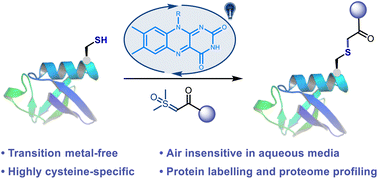
Visible-light-mediated methods were heavily studied as a useful tool for cysteine-selective bio-conjugation; however, many current methods suffer from bio-incompatible reaction conditions and slow kinetics. To address these challenges, herein, we report a transition metal-free thiol-sulfoxonium ylide photo-click reaction that enables bioconjugation under bio-compatible conditions. The reaction is highly cysteine-selective and generally finished within minutes with naturally occurring riboflavin derivatives as organic photocatalysts. The catalysts and substrates are readily accessible and bench stable and have satisfactory water solubility. As a proof-of-concept study, the reaction was smoothly applied in chemo-proteomic analysis, which provides efficient tools to explore the druggable content of the human proteome.


 Please wait while we load your content...
Please wait while we load your content...