An iron-based metal–organic framework for selective CO2 adsorption and as an efficient anode material for lithium-ion batteries†
Abstract
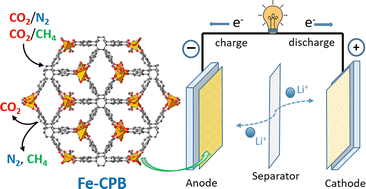
Given the versatility of electronic properties induced by the synergic effect of metal-oxo clusters and multifunctional organic ligands, metal–organic frameworks (MOFs) have emerged as prospective materials for energy-related applications such as gas storage/separation and electrochemical storage. In this study, an iron-based MOF, namely, Fe-CPB (CPB = 1,2,3,4,5,6-hexakis(4-carboxyphenyl)benzene) was successfully synthesized via a solvothermal reaction and fully characterized. Taking advantage of the permanent porosity, the layered structure with high π-conjugation derived from a hexaphenylbenzene core, and polar carboxylate and acetate groups connected by infinite iron-oxo chains, Fe-CPB has been examined for its selective CO2 adsorption properties and electrochemical performance when applied as an anode material for lithium-ion batteries. As a proof-of-concept, Fe-CPB showed a moderate low-pressure CO2 uptake (40 cm3 g−1 at 273 K), high isosteric heat of CO2 adsorption (33 kJ mol−1), and good CO2 selectivity over N2 and CH4. As an anode material for rechargeable lithium-ion batteries, Fe-CPB exhibited a high reversible capacity of 634 mA h g−1 with 99% capacity retention after 100 cycles (at a current density of 100 mA g−1). Interestingly, Fe-CPB showed a high rate capability with a discharge capacity of 416 mA h g−1 (at a current density of 2000 mA g−1) and 673 mA h g−1 when the current density was returned to 100 mA g−1. Our result envisaged the potential of developing MOFs for energy applications including CO2 separation and electrodes of rechargeable batteries.


 Please wait while we load your content...
Please wait while we load your content...