Soluble sulfonated polybenzimidazoles containing phosphine oxide units as proton exchange membranes†
Abstract
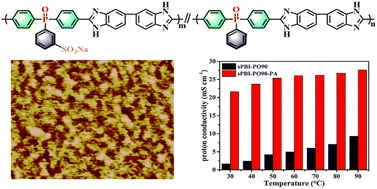
Sulfonated bis(4-methyl benzoate) phenylphosphine oxide (sBMPO) was synthesized by the sulfonation and esterification of bis(4-methylbenzoate)phenylphosphine oxide (BMPO). A series of high-molecular-weight sulfonated polybenzimidazoles (sPBI-PO) was also prepared via the random co-polycondensation of sBMPO, BMPO, and 3,3′-diaminobenzidine. The solubility was improved by incorporating a high concentration of phenylphosphine oxide units with pendant benzene groups in the polymers due to the remodeling of an irregular arrangement of polymer main chains. The sPBI-PO polymers showed excellent mechanical properties and thermal stability. The prepared sPBI-PO membranes displayed suitable water uptake and excellent dimensional stability in a very narrow range of 7.4–9.5% because of the good water retention ability of the phenylphosphine oxide units. With increasing sulfonation degree, the hydrophilic/hydrophobic nanophase separation morphology of sPBI-PO became more obvious, as shown by AFM phase images. The sPBI-PO membranes exhibited relatively low proton conductivity due to the strong acid–base interaction between the sulfonated group and the benzimidazole ring. However, after doping with phosphoric acid, the proton conductivity of the sPBI-PO90-PA membrane was 27.62 mS cm−1, which was 3.89 times higher than that of the undoped sPBI-PO90 membrane (7.1 mS cm−1) at 80 °C and 100% RH. Hence, these sPBI-PO polymers are highly promising matrix materials for PEMs.


 Please wait while we load your content...
Please wait while we load your content...